Caption
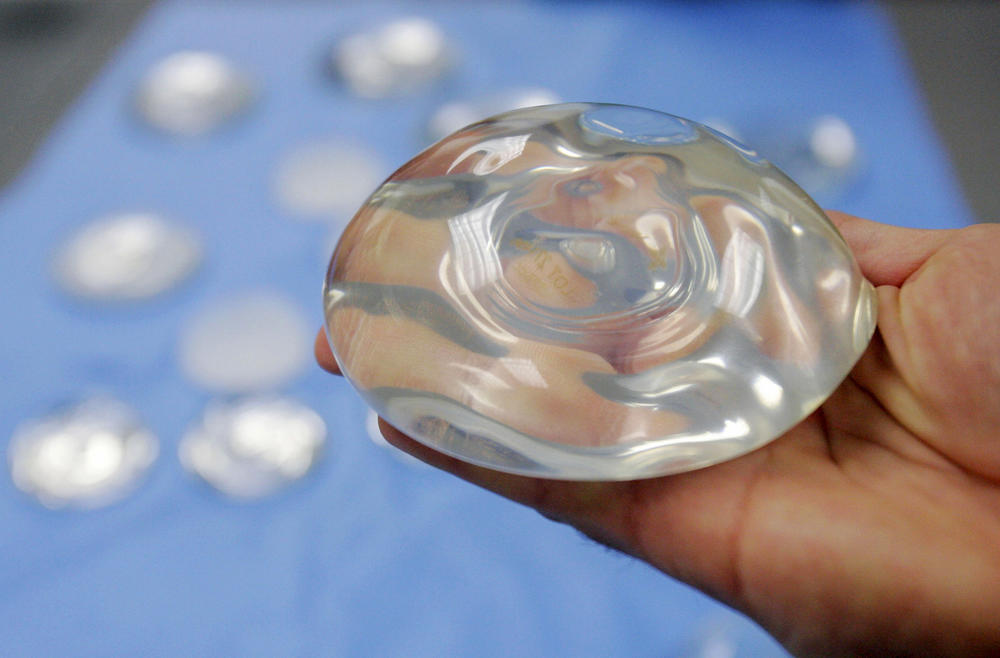
In this Dec. 11, 2006, file photo, a silicone gel breast implant is shown at Mentor Corp., a subsidiary of Johnson & Johnson, in Irving, Texas.
Credit: (AP Photo/Donna McWilliam, File)
|Updated: October 7, 2020 4:37 AM
An Atlanta plastic surgeon is recruiting patients to study the impact of breast implants. This comes after the U.S. Food and Drug Administration issues its strongest warning yet for all breast implant products. GPB’s Ellen Eldridge reports.

In this Dec. 11, 2006, file photo, a silicone gel breast implant is shown at Mentor Corp., a subsidiary of Johnson & Johnson, in Irving, Texas.
The U.S. Food and Drug Administration last week issued its strongest warning for all breast implant products.
The FDA’s labeling guidance comes 18 months after an advisory panel heard from dozens of women who say their breast implants made them sick.
PREVIOUS COVERAGE: Illness Caused By Breast Implants, Georgia Women, Plastic Surgeons Tell FDA
They say they were never told implants are not a lifetime device.
Dr. Marisa Lawrence is an Atlanta-based plastic surgeon who says implants are associated with cancer of the immune system, anaplastic large cell lymphoma (BIA-ALCL).
The risk of developing BIA-ALCL is considered low, but the cancer is serious and can lead to death, according to the FDA.
The black box warning is an important improvement for breast implant safety, she said.
"That's the strongest warning by the FDA that a product carries the risk of serious injury or death," Lawrence said.
Patients have reported systemic symptoms such as joint pain, muscle aches, confusion, chronic fatigue and autoimmune diseases that improve when the implants are removed, but the FDA's warning does not specifically mention the term breast implant illness.
Lawrence is conducting research about the causes of systemic illness associated with breast implants.
She said in her 27-year career as a plastic surgeon the medical community has never seen evidence linking breast implants to any disease. There is also no proof implants are safe.
"The first round of research back in the '90s did not give us the data or the research they wanted," she said.
Then, in 2016 and 2017 when patients again complained of systemic illness associated with breast implants, the manufacturers and doctors again asked for proof.
"The medical community from the get-go has said, 'Show us the scientific data. Show us the literature. We need scientific evidence,'" Lawrence said.
Lawrence first started with a questionnaire asking her patients about their symptoms, which type of breast implants patients had and how long they had implants before experiencing symptoms.
Then, her plastic surgery group received the largest grant ever from the Aesthetic Surgery Education Research Foundation to do research including DNA evaluation of the capsule tissue.
"We have a toxicologist, we have a pathologist. We have a microbiologist, all top in their field, working with my group of surgeons, looking at this, trying to figure out exactly what's the cause of breast implant illness," she said.
While the pandemic has slowed the study a bit, Lawrence said her group got back on track in June.
"We are continuing to enroll patients and we're hoping to have complete enrollment by the end of November," she said. "And we'd love to get preliminary results in by January."