Section Branding
Header Content
Emory Seeks Clinical Trial Participants For COVID-19 Vaccine
Primary Content
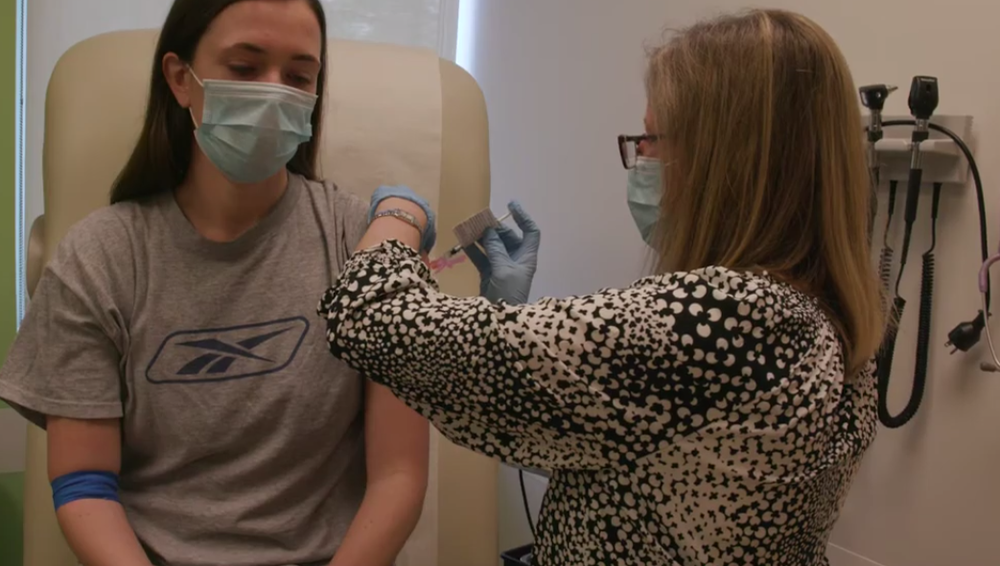
Atlanta's Emory University is enrolling participants for the nation's first clinical trial for a COVID-19 vaccine.
Emory's Vaccine and Treatment Evaluation Unit is testing "mRNA-1273," which was developed by the National Institute of Allergy and Infectious Diseases and Moderna Incorporated. Phase I of the study began March 16 at the VTEU at the Kaiser Permanente Washington Health Research Institute in Seattle.
The trial hopes to recruit 45 people between the two locations. Participants must be adults in the Atlanta area age 18 to 55. To be eligible, they can’t have chronic diseases or health conditions that affect the immune system, and they can’t be taking immunosuppressive medications.
Other criteria and additional information about the vaccine study are available at ClinicalTrials.gov.
MORE: Worried You Have COVID-19? Emory Has A Website To Check
Leading the trial at Emory is Associate Professor of Medicine and Pediatrics Dr. Evan Anderson and interim Director of the Hope Clinic of the Emory Vaccine Center Nadine Rouphael.
Rouphael said the team is ready for the challenge of creating a vaccine.
“A vaccine against COVID-19 is urgently needed because of widespread infection and lack of preexisting immunity,” Rouphael said. “We are looking forward to being part of a nationwide effort to respond to this crisis.”
RELATED: Emory Law Student: 'How Are We Supposed To Have A Bar Exam?'
One of the volunteers for the vaccine study, Claire MacDougall, said she participated because she felt a duty to help others during the pandemic crisis.
"In the back of my mind, I could do something to help the entire world by doing this," MacDougall said. "There isn't one person that isn't affected by this right now."
If the trial goes well, a vaccine could be available to the public in a little over a year, Anderson said.
MORE: University Of Georgia Works To Develop A Vaccine for Coronavirus